New Mesothelioma Drug Tests to Begin Soon
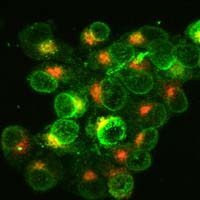
A spokesman for the makers of a promising new mesothelioma drug says the company plans to begin enrolling its first clinical trial participants this summer. Dr. Joanna Horobin is Chief Medical Officer for Verastem, Inc., the developer of a drug that aims to treat malignant mesothelioma by targeting the stem cells that give rise to it. The company’s lead compound, an oral drug called VS-6063, inhibits a crucial signaling pathway inside stem cells called the Focal Adhesion Kinase (FAK) pathway. VS-6063 was approved by the FDA earlier this year as a ‘orphan drug’, a designation given to drugs designed to treat rare illnesses like mesothelioma. “We are moving quickly to bring new treatment options to patients with mesothelioma,” Dr. Horobin said…