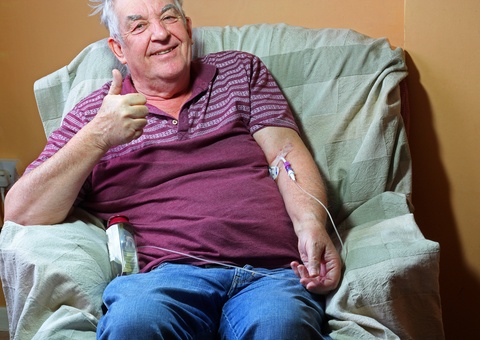
Faster Mesothelioma Chemotherapy Deemed “Safe and Feasible”
Researchers in The Netherlands say it may be possible to deliver faster mesothelioma chemotherapy without doing any additional damage to the kidneys. If they are right, it could make mesothelioma treatment more accessible and convenient for patients. Most malignant mesothelioma patients will have chemotherapy. The most common type of chemotherapy is by infusion. Each session takes several hours. Every mesothelioma patient undergoing chemotherapy faces the possibility of kidney damage. That possibility can even limit how much medicine doctors can use. But a new Dutch study finds that faster mesothelioma chemotherapy may be as safe for the kidneys as standard chemotherapy. Analyzing Faster Mesothelioma Chemotherapy The study included 230 lung cancer patients who had chemotherapy. Some of the study subjects had…